Innovative Micro-porous Material Offers New Carbon Capture Options
A relatively new class of engineered materials known as metal-organic frameworks (MOFs) may provide an effective alternative for capturing carbon dioxide (CO2) from fossil fuel combustion emissions, which makes it a significant new option in emissions reductions.
#materials
THE SITUATION
Power plants burning fossil fuels account for over 2 billion tons (1.8 billion metric tons) of CO2 annually, which is 37% of all U.S. emissions, according to a report from University of California, Berkeley. An essential factor in looking for ways to reduce emissions is carbon capture and storage (CCS).
Removing CO2 from flue gases (called post-combustion carbon capture) is a significant area of research as it could be easily retrofitted to existing plants. However, this technique is technically challenging and can be energy intensive, which reduces the overall efficiency of power production, the report said.
Companies, universities and government agencies continue to explore new technologies for carbon capture and storage, and many different methods could do the job for different situations. However, the challenge is in finding those that can do it efficiently and affordably.
Yuzhang Li, a Stanford University postdoctoral researcher said in a report on MOFs that: “According to the Intergovernmental Panel on Climate Change, limiting global temperature increases to 1.5⁰C [2.7⁰F] will require some form of carbon capture technology. These materials have the potential to capture large quantities of CO2” and may allow carbon capture that is more efficient and less expensive than existing technologies.
METAL-ORGANIC FRAMEWORK AS A SOLUTION
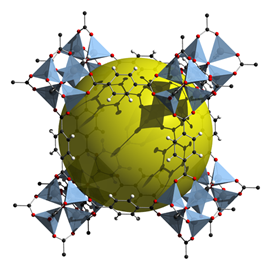
An MOF is a hybrid organic-inorganic crystalline, porous material. Its structure is a regular array of positively charged metal ions surrounded by organic, linking molecules. The metal ions bind the links together to form a three-dimensional, repeating cage structure, Figure 1. This hollow matrix configuration gives MOFs enormous surface area per volume and the ability to capture molecules of the appropriate size. Some MOFs achieve a surface area of more than 7,000 square meters (more than 75,000 square feet) per gram (0.035 oz). This is equivalent to a teaspoonful of the material having surface area greater than the area of an American football field.
Because MOFs can be produced with various combinations of metal ions and organic links to create different sizes and geometries of pores, they can be tailored to the size of whichever molecules need to be captured. In addition to CO2 capture, prospective uses for MOFs include separation and purification of gases, catalyst applications and storage of fuels such as hydrogen (Figure 2) or methane.
MOFS FOR CARBON CAPTURE
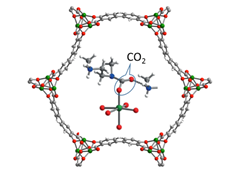
For carbon capture, MOF properties are fine-tuned to “selectively bind CO2 with minimum energy cost and to withstand the harsh post-combustion environment,” according to the UC Berkeley report (Figure 3).
In addition to its ability to effectively capture CO2 molecules, the MOF approach allows release of the CO2 by a relatively low-energy process. This release process is referred to as regeneration. It allows the CO2 to be collected for storage or to be used in other processes, such as oil extraction enhancement. After regeneration, the MOF is ready to capture more CO2.
PUTTING CARBON CAPTURE INTO PRACTICE
Options for CO2 capture from flue gases range from various kinds of solvent processes to using membranes or enzyme processes. Another approach is pre-combustion carbon reduction, which mainly relates to gasifying the fuel so it has lower carbon content and produces less carbon dioxide when burned or processed in a fuel cell.
Though much research has been done on post-combustion carbon capture technologies, few large-scale projects are currently online. These CCS installations, such as the Petra Nova project at a coal-fired power plant near Houston, generally capture CO2 by absorbing it in a solvent. MOF technologies have the potential for enabling economical and scalable carbon capture. A recently initiated project explores using the method to meet increased energy demands while reducing emissions. The two participants are ExxonMobil and Mosaic Materials, a technology company that is commercializing MOF technology originally developed at the University of California, Berkeley.
The main challenges in bringing new CCS technologies to practical use according to the UC Berkeley report are:
- demonstration at the scale necessary for power plant application
- reduction of the parasitic loads (steam and power) required to support CO2 capture that significantly decrease power generating capacity
- reduction of the costs of scaled-up processes to minimize the impact on the cost of electricity.
The use of MOFs “holds promise for cost-effective carbon capture in the future, as well as energy-efficient applications in related industrial processes,” said the UC Berkeley report. Testing continues to rapidly screen potentially useful MOF compounds under conditions mimicking flue gas situations in power plants. The result may be further improvements in MOF-related innovative technologies for industrial carbon capture that provide improvements in cost and performance.
Barbara Donohue is web editor at VALVE Magazine
RELATED CONTENT
-
PFAS Chemicals and PTFE: Should the Valve Industry Be Concerned?
Legislation moving through Congress could affect the future use of thousands of PFAS chemicals (per- and polyfluoroalkyl). The house passed H.R. 2467 in July of 2021 and, though the bill is general in nature, it assigns the responsibility to the Environmental Protection Agency (EPA) for determining which PFAS chemicals will be controlled or banned altogether.
-
The Materials That Make Up Valves
EDITOR’S NOTE: Materials used in the manufacture of valves and how they perform in different applications is a topic of huge interest to everyone who works with valves.
-
Hardfacing for Valves: Materials and Processes
Valve internals, such as seats and closures, are often at risk of erosion, abrasion, corrosion, galling and damage from cavitation.









 Unloading large gate valve.jpg;maxWidth=214)